Background
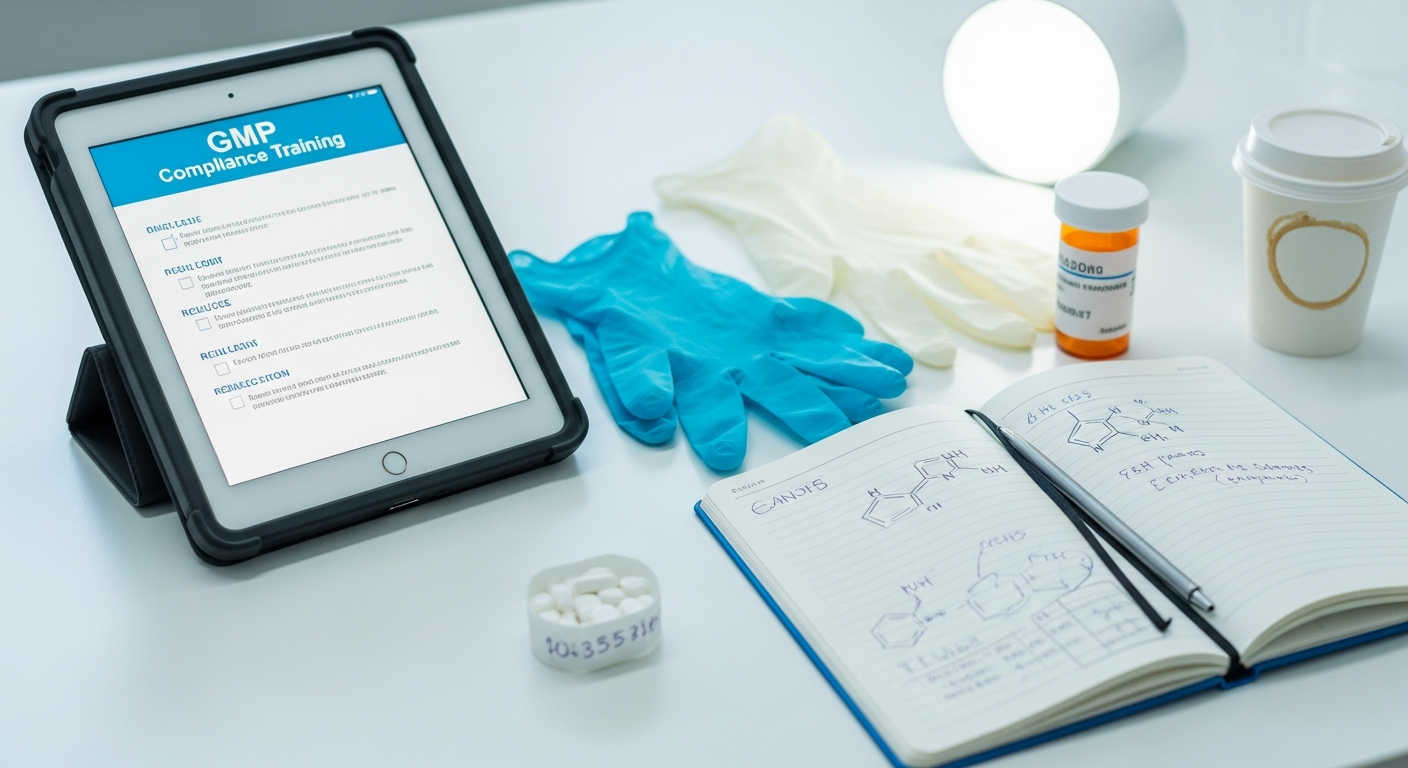
Production floor employees must regularly complete mandatory training — GMP, pharmaceutical safety, quality management. High deskless worker ratios and shift schedules made classroom attendance difficult, and PC access on the production line was limited. Compliance tracking consumed significant administrative resources.
Solution
Applied P3 to convert GMP and safety training to mobile with push notifications and non-completer targeting, aligned to shift schedules. Expanded from compliance into P4 (social learning): production workers shared quality issue cases and safety prevention tips on social boards, maintaining engagement reasons beyond compliance windows.
Results
Achieved compliance MAU consistent with the P3 median of 89% (range 78–97%). Mobile reach to field workers reduced compliance tracking admin costs. Social board knowledge sharing activated post-compliance, sustaining the platform as an always-on channel.
Insight
The observation that in manufacturing/logistics, mobile accessibility itself is the prerequisite for non-desk workers applies equally to medical-device and pharma companies. The industry MAU comparison puts manufacturing/logistics (n=5) at a median of 22% — the lowest — precisely because of this access constraint. Using mandatory training to form an access habit and social learning to sustain it is the dual-structure L&D design principle for pharma/medical-device field operations.